 The fact that a perfect crystal of a substance at 0 K has zero entropy is sometimes called the Third Law of Thermodynamics. If the only work done is a change of volume at. According to the law of energy conservation, the change in internal energy is equal to the heat transferred to, less the work done by, the system. This is because we know that the substance has zero entropy as a perfect crystal at 0 K there is no comparable zero for enthalpy. In symbols, the enthalpy, H, equals the sum of the internal energy, E, and the product of the pressure, P, and volume, V, of the system: H E + PV. 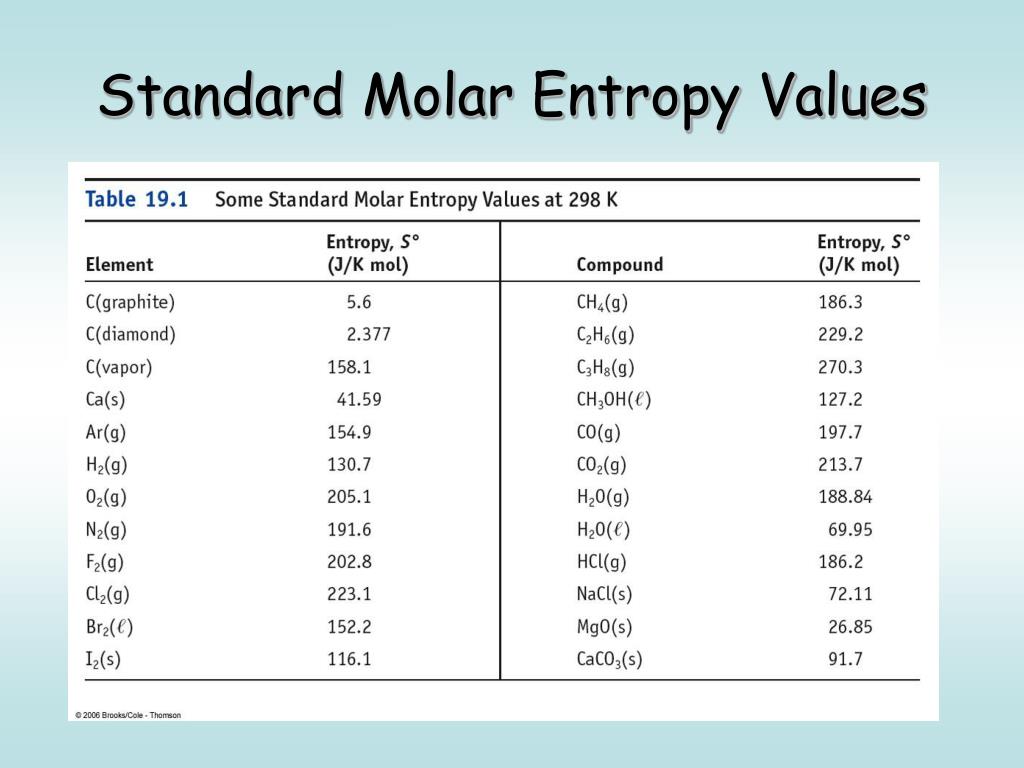
The reason is that the entropies listed are absolute, rather than relative to some arbitrary standard like enthalpy. Note that there are values listed for elements, unlike DH fº values for elements. It equals to the total entropy (S) divided by the total mass (m). The specific entropy (s) of a substance is its entropy per unit mass. Engineers use the specific entropy in thermodynamic analysis more than the entropy itself. The Thermodynamics Table lists the entropies of some substances at 25 ✬. The entropy can be made into an intensive, or specific, variable by dividing by the mass. Continue this process until you reach the temperature for which you want to know the entropy of a substance (25 ✬ is a common temperature for reporting the entropy of a substance). For standard entropy, this assumes that the. Then you can use equation (1) to calculate the entropy changes. The units of entropy are JK-1mol-1, which basically means joules of energy per unit heat (in Kelvin) per mol. Even though equation (1) only works when the temperature is constant, it is approximately correct when the temperature change is small. For processes involving an increase in the number of microstates, Wf > Wi, the entropy of the system increases and S > 0. Now start introducing small amounts of heat and measuring the temperature change. As for other state functions, the change in entropy for a process is the difference between its final ( Sf) and initial ( Si) values: S S f S i k ln W f k ln W i k ln W f W i. Since there is no disorder in this state, the entropy can be defined as zero. 
Imagine cooling the substance to absolute zero and forming a perfect crystal (no holes, all the atoms in their exact place in the crystal lattice). The absolute entropy of any substance can be calculated using equation (1) in the following way. At absolute 0 (0 K), all atomic motion ceases and the disorder in a substance is zero. Originally defined in thermodynamics in terms of heat and temperature, entropy indicates the degree to which a given quantity of thermal energy is available for. On this scale, zero is the theoretically lowest possible temperature that any substance can reach. As the following example demonstrates, entropy is a property of a system. The temperature in this equation must be measured on the absolute, or Kelvin temperature scale. The units of entropy and entropy change are joules per kelvin (J/K). Using this equation it is possible to measure entropy changes using a calorimeter. 
Where S represents entropy, DS represents the change in entropy, q represents heat transfer, and T is the temperature. Entropy quantifies the energy of a substance that is no longer available to perform useful work. where: s specific entropy (J/kg) S entropy (J) m mass (kg) T-s diagram of Rankine Cycle. In all other cases the system (the gas) is restored to its initial state, but the surroundings are forever changed.Ī reversible change is one carried out in such as way that, when undone, both the system and surroundings (that is, the world) remain unchanged.One useful way of measuring entropy is by the following equation: The specific entropy (s) of a substance is its entropy per unit mass. In the limit of an ”infinite” number of steps (bottom), these work terms are identical, and both the system and surroundings (the “world”) are unchanged by the expansion-compression cycle. 
As the number of steps increases, the processes become less irreversible that is, the difference between the work done in expansion and that required to re-compress the gas diminishes. The thing you must be most careful about is the fact that entropy is measured in joules, not kilojoules, unlike most of. These diagrams show the same expansion and compression ±ΔV carried out in different numbers of steps ranging from a single step at the top to an "infinite" number of steps at the bottom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
